Actuate Therapeutics seeks IPO for promising pancreatic cancer drug (Pending: ACTU)
ljubaphoto/E+ via Getty Images
Actuate Therapeutics, Inc. at a glance
Activate Therapeutics, Inc (ACTU) has filed to raise $50 million in an IPO of its common stock, according to its SEC S-1 filing information.
The company is clinical stage The biopharmaceutical company develops treatments for pancreatic cancer and other cancers.
Actuate Therapeutics, Inc. has demonstrated Promising efficacy results in phase 2 trials of its flagship program.
I will provide a final opinion when we learn more about the IPO.
Activate overview
Actuate Therapeutics, Inc. was founded. in Fort Worth, Texas, to license technologies called GSK-3 inhibitors that promise to block the function of the enzyme GSK-3B, “a master regulator of complex biological signaling cascades…that lead to tumor.” Cell survival, migration, growth, and invasion.”
Management is led by President and CEO Daniel M. Schmidt, who has been with the company since March 2015 and has been He was previously Chief Operating Officer of Genus Oncology and has extensive experience in the pharmaceutical industry.
The company’s lead drug candidate is elraglusib, which is being developed to treat several cancer conditions.
Its pancreatic cancer program is the most advanced, having completed Phase 1/2 trials, and the Department is continuing to test the treatment for Ewing’s sarcoma as part of the “Phase 1/2 Actuate-1902 study in refractory pediatric malignancies.”
The current status of the company’s development pipeline is shown here:
second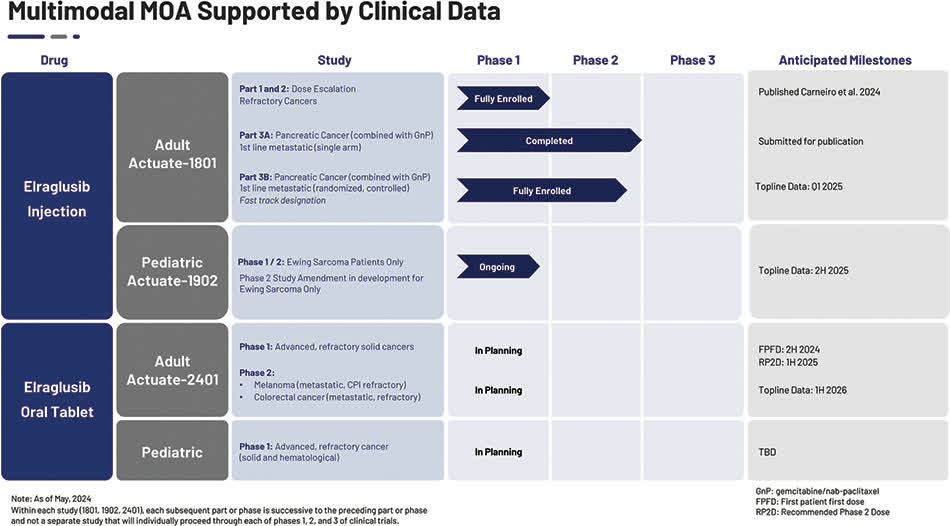
Actuate has booked an investment with a fair market value of $99.8 million as of March 31, 2024, from investors, including Bios Equity affiliated funds and Kairos Venture affiliated funds.
Market and competition in Actuate
According to a 2019 market research report by Grand View Research, the global pancreatic cancer treatment market was estimated at $560 million in 2015, and is expected to reach $4.2 billion by 2025.
This represents an expected compound annual growth rate (CAGR) of 23% from 2016 to 2025.
The main factors driving this expected growth are the growing elderly population worldwide, which leads to decreased immune system function, a higher incidence of cancer, and a higher prevalence of alcohol consumption and obesity.
The chart below also shows the historical and expected future growth of the pancreatic cancer treatment market worldwide until 2025:
Grand View Research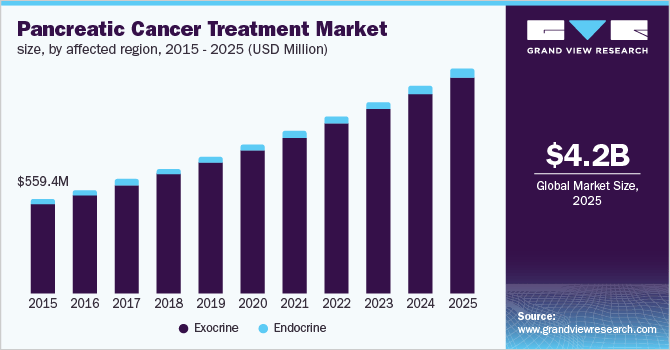
Key competitive vendors that have developed or are developing similar treatments include the following companies:
-
Eli Lilly & Company
-
Celgene
-
F. Hoffmann-La Roche AG
-
Amgen
-
Novartis
-
Pharmaset Biotechnology
-
Clovis tumors
-
Teva Pharmaceutical
-
Merck & Co
-
Pfizer
-
Others.
Activating the therapeutic financial situation
The company’s recent financial results are typical for clinical-stage biopharmaceutical companies as they do not contain any of the significant R&D, general and administrative revenues and expenses incurred by various development programs.
Below are the company’s current financial results over the past two calendar years, according to its most recent IPO filing documents:
second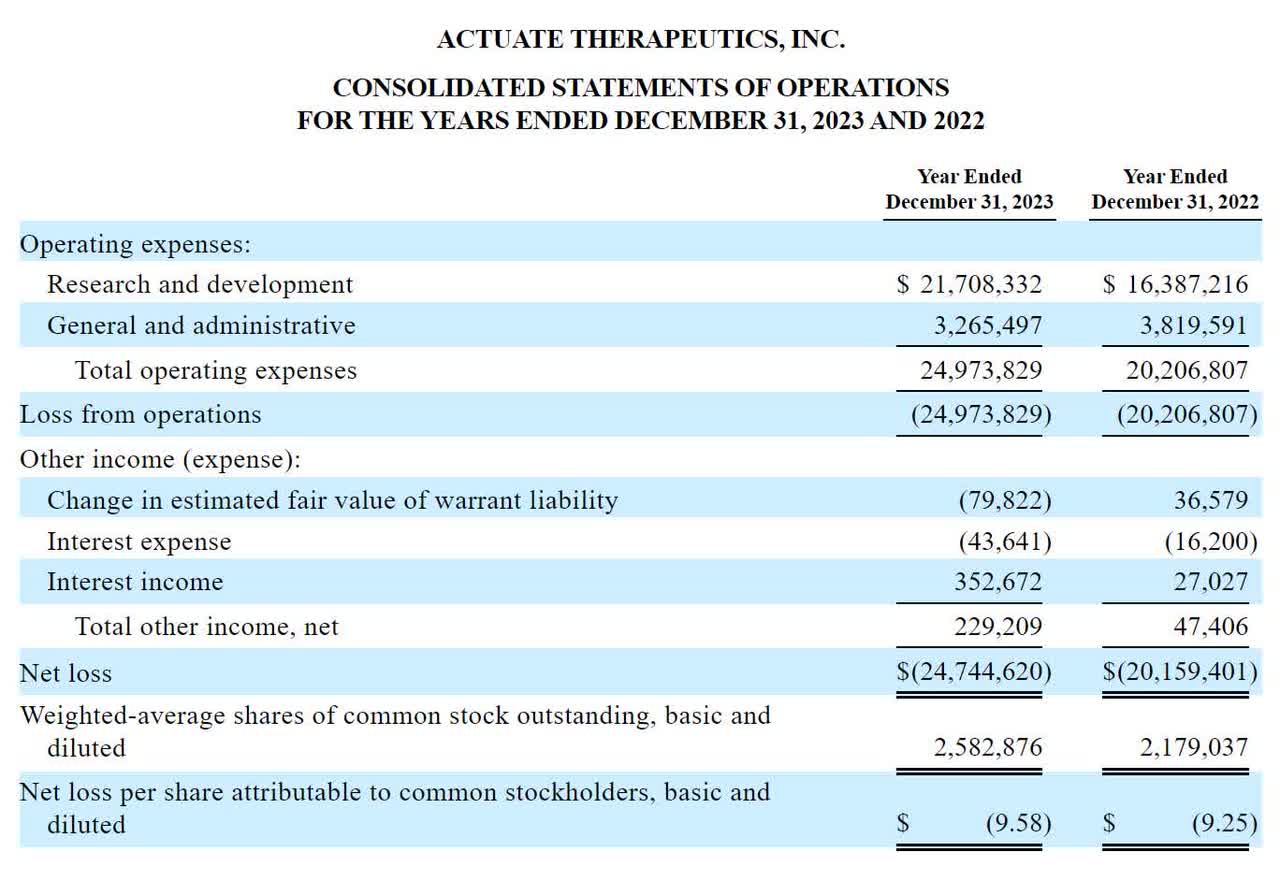
As of March 31, 2024, the company had $2.1 million in cash and $16.3 million in total liabilities.
Activate details of Therapeutics’ IPO
Actuate intends to raise $50 million in gross proceeds from an initial public offering of its common stock, although the final number may be higher.
No current or potential shareholders have shown interest in acquiring shares at the IPO price.
The company has elected to be an “emerging growth company” and a “smaller reporting company” in accordance with the JOBS Act of 2012.
These classifications enable management to provide much less information to shareholders at their discretion.
Given the development stage of the company’s pipeline, I estimate that Actuate will have a market capitalization at IPO of approximately $200 million.
Management said it plans to use the net proceeds from the IPO as detailed here:
Ongoing mPDAC phase II Elraglusib injection trial (Actuate-1801 Part 3B)
Funding the current phase I dose escalation trial for refractory pediatric cancer and further exploring development opportunities and potentially initiating the second part of this study in patients with refractory Ewing sarcoma (Actuate-1902)
Meet the company’s funding commitments for ongoing IIT studies of the use of Elraglusib injection in combination with other chemotherapy agents to treat mPDAC and a separate trial for the treatment of recurrent salivary gland carcinoma.
To the extent any proceeds remain available, fund the initial planning steps for the studies described below and additional general and administrative operations.
(Source: SEC.)
The online driving company promotion is not yet available.
Regarding the legal proceedings, management said that the company is not a party to any legal claims that, if brought against the company in their entirety, would have a material adverse effect on its financial condition or operations.
Bookrunners listed on the IPO are Titan Partners Group and Newbridge Securities.
Comment on Actuate’s IPO
ACTU is seeking public capital market investment in the US to develop its Elraglusib treatment for various cancers.
Its pancreatic cancer program is the most advanced, having completed Phase 1/2 trials, and management is continuing to test the treatment for Ewing’s sarcoma as part of the Phase 1/2 Actuate-1902 study in refractory pediatric malignancies.
The market opportunity for treating pancreatic cancer is fairly large, but it has proven difficult to develop effective treatments for this condition, which is difficult to detect at an early enough stage to produce successful outcomes.
Actuate is developing its lead candidate for the treatment of other cancers, which have reasonably large markets that are also growing due to aging populations and decreased immune system response capabilities.
The administration did not reveal any major agreements or cooperation relationships with pharmaceutical companies.
The company’s investor syndicate includes investors from venture capital firms focused on biotechnology.
Actuate has demonstrated preliminary efficacy results for the second phase of its flagship program, as management states here:
Based on these interim data, the preliminary Kaplan-Meier analysis shows a median overall survival (mOS) of 12.2 months in the elraglusib combination arm versus 7.3 months in the GnP control arm (HR=0.60; log-rank p=0.012). .
So, ACTU appears to have phase II trial activity that is very promising for this cancer condition that has proven difficult to treat.
As we learn management’s assumptions about IPO pricing and valuation, I will provide an update.
Expected IPO pricing date: To be announced.





