BridgeBio Pharma: Strengthens Existing Position (Rating Upgrade) (NASDAQ:BBIO)

anusorn nakdee
When we last looked Bridge Bio Pharma Inc. (Nasdaq: Pebio) Going back to early December of last year, the stock has recently doubled. Our advice then condition Had to take at least some profits, shares They seem to have gotten a little ahead of themselves.
Seeking alpha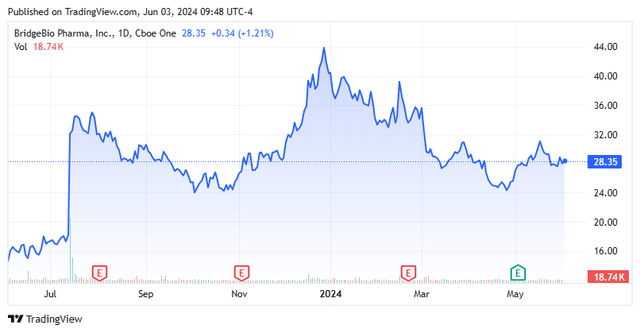
The stock is down about 10% since we published this research. Wells Fargo recently named BridgeBio Pharma after it existing A potential target for a biotechnology buyout, the company has published some additional trials results At the end of May for lead drug candidate Acoramidis. Therefore, it seems like a good time to return to mid-sized biotech concerns. An updated analysis follows below.
BridgeBio is headquartered in Palo Alto, California. The company has somewhat of a unique business model in the industry, operating a hub and spoke network of Affiliate Companies Which works to develop treatments for individual diseases. From that network, the company has dozens of candidates in both pre-clinical and clinical development. The stock is currently trading at around $29.00 per share and has an approximate market cap of $5.3 billion.
The most important BridgeBio asset in development is called Acoramidis as mentioned earlier. This product has a marketing application that has been accepted by the US Food and Drug Administration (FDA) for review, with a decision scheduled for late November of this year on whether to approve Acoramidis for the treatment of amyloid transthyretin cardiomyopathy, or ATTR-CM. A decision on approval in Europe is scheduled for the first half of 2025. Acoramidis is also known as AG10 and is an investigational next-generation transthyretin stabilizer.
It is worth noting that Pfizer (PFE) It contains a compound called tafamidis that is approved for ATTR-CM and is marketed under the brand names Vyndaqel (Europe) and Vyndamax (US). This compound Delivered Global sales were $1.1 billion in the first quarter, well above analysts’ expectations of $900 million. Pfizer is working feverishly to expand patent protection for this drug, which is set to expire in the United States later this year and in 2026 in Europe.
Mars Company Presentation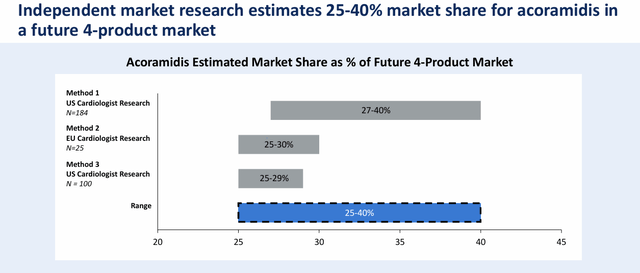
A recent analysis conducted via another “Seeking Alpha”. condition It predicted earlier this year that Acoramidis could eventually generate between $3.75 billion and $6 billion at peak sales. This forecast took into account management’s goal of capturing 25% to 40% of ATTR-CM, and growing this market to $15 billion globally. There are other potential ATTR-CM treatments in the pipeline Ionis Pharmaceuticals Company (ions), AstraZeneca Limited Liability Company (AZN), And Al-Naylam Pharmaceutical Company (Allen).
Pfizer presentation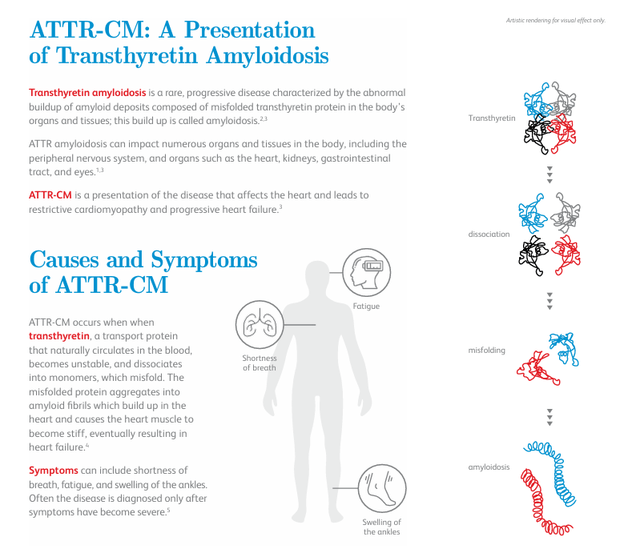
Bridge Bio, as previously mentioned, has dozens of candidates in some form of development across its corporate pool (17 INDs overall). Management provided an update on progress on its most important programs during the first quarter press release. This analysis will focus primarily on the development of Acoramidis. A good view of BridgeBio’s extensive pipeline can be seen here.
May 2024 Company Presentation
Analyst view of the company and balance sheet status:
It’s worth noting that the analyst firm community is globally positive about BridgeBio’s prospects. Since the company published First quarter results In early May, dozens of analyst firms, including UBS, Wells Fargo, and Citigroup, reissued/assigned buy/outperform ratings on BBIO. It should be noted that eight of these rankings have been issued since the latest trial data appeared last week. Price targets offered range from $43 to $58 per share.
March 2024 Company Presentation
The company ended the first quarter with approximately $520 million of cash and marketable securities on its balance sheet. In a show of confidence, Bayer Inky A deal with BridgeBio to acquire the marketing rights to Acoramidis in Europe in March this year. Bayer will pay US$310 million as an upfront payment and near-term milestone payments. BridgeBio will also be eligible for additional sales payments, as well as tiered royalties starting in the low 30s on commercial sales of Acoramidis in Europe.
The company too executed Of note is a secondary offering in early March that raised approximately $287 million. Finally, BridgeBio will receive half a billion dollars from Blue Owl Capital and the Canada Pension Plan Investment Board following the US Food and Drug Administration’s approval of Acoramidis in exchange for five percent of the drug’s global net sales.
Conclusion:
The latest phase 3 data for Acoramidis ATTR-CM published on Wednesday appears to have fueled investor enthusiasm about the potential approval of Acoramidis in November, with shares rising about seven percent since then as several analyst firms chimed in with “excellent.”
Mars Company Presentation
Given the potential approval of Acoramidis and its potential peak sales against the company’s market capitalization of just over $5 billion, I believe it is time to establish an additional position in BBIO via covered call orders. That’s what I did on Monday to start the new trading month in June.
Finally, it’s also worth noting that my favorite long-term ATTR-CM play is my interest in gene editing Intellia Therapeutics Inc (NTLA) Published by A Deep dive In late February. “It has potential”therapeutic” Single dose candidate for this disease. However, the product is likely to hit the market in 2027 at the earliest.





