Revolution Drugs: Waiting for a Lower Entry Point (NASDAQ:RVMD)

Sangiri
Shares of oncology concern Al-Thawra Pharmaceuticals Company (Nasdaq: RVMD) has more than doubled since the market’s initially negative reaction to the clinical update of lead asset RMC-6236 in October 2023. The company’s new approach to treating cancer with Its RAS(ON) inhibitors will be tested when RMC-6236 enters two registrational trials for pancreatic and lung cancer at the 22nd hour. With plenty of catalysts to come, an operating runway to 2027, and being listed as a potential acquisition target by Wells Fargo, Revolution is worth revisiting. An updated analysis follows in the paragraphs below.
Seeking alpha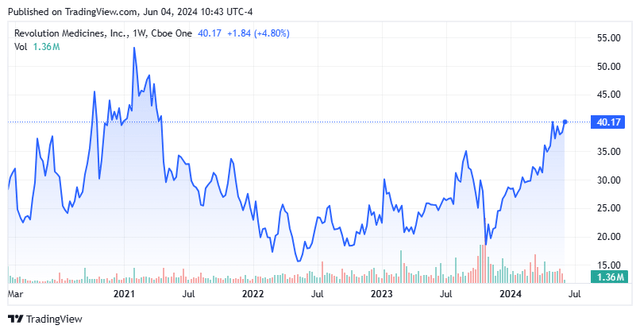
when we finished seem In the company for more than 14 months, it had ended a five-year collaboration with Sanofi and recently implemented a spin-off. Its shares were trading in the low 20s and a small position was recommended based on its new approach to cancer treatment With RAS(ON) inhibitors, supported by supportive early clinical returns.
As a reminder, Revolution Medicines, Inc. is a clinical-stage precision oncology company in Redwood City, California, focused on developing drugs that inhibit frontier targets of cancers that are dependent on the renin-angiotensin system (RAS). The company is currently developing three clinical programs – all novel RAS(ON) inhibitors – against solid tumor targets. Revolution was founded in 2014 and went public in 2020, raising net proceeds of $251.0 million at $17 per share. The stock is trading at around $40.00 per share, which translates to an approximate market capitalization of $6.6 billion.
RAS inhibition
RAS controls systemic body fluid circulation, but is also present in some tumor microenvironments. In healthy patients, the RAS acts like a light switch, toggling between “ON” and “OFF.” When turned on, the RAS isoform protein sends signals that tell the cell to grow and divide. Mutated RAS proteins are rarely turned off, leading to uncontrolled cell growth. Once considered”Not druggable“, treatments that attempt to keep the RAS permanently locked in the off mode have been approved, starting with Amgen (AMGN) KRAS G12C (OFF) inhibitor Lumakras (sotorasib) in 2021. By contrast, Revolution’s leading treatments aim to permanently unplug the ON switch.
The company has two types of RAS-targeting compounds: the RAS inhibitors (ON) just described, which aim to block the ON switch by inhibiting active RAS bound to guanosine triphosphate (or ON form) near the cancer mutation’s ovules (central points). ) and suppress excessive signaling at its source, and concomitant RAS inhibitors, which suppress cooperating targets and pathways that underpin RAS-addicted cancers and are therefore designed to be used in combination with other therapies to counter treatment resistance. Excessive RAS (ON) signaling accounts for approximately 30% of cancer cases, including approximately 60,000 new cases of lung cancer, approximately 75,000 new diagnoses of colorectal cancer, and approximately 53,000 new cases of pancreatic cancer in the United States annually. Given the results achieved in the clinic to date, the company’s focus has shifted almost exclusively to RAS inhibitors (ON).
May 2024 Company Presentation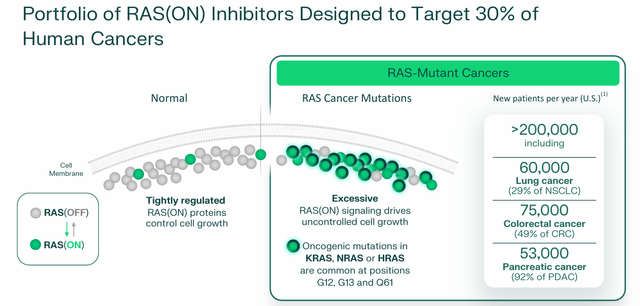
Pipeline
RMC-6236. Revolution’s gaudy market capitalization of $6.22 billion is largely attributable to the company’s flagship program, RMC-6236, which is designed to prevent a spectrum of RAS (ON) variants, including all three major RAS isoforms (KRAS, NRAS, and HRAS). ) that are found in the mutation hotspot positions G12 (most prevalent), G13, and Q61.
May 2024 Company Presentation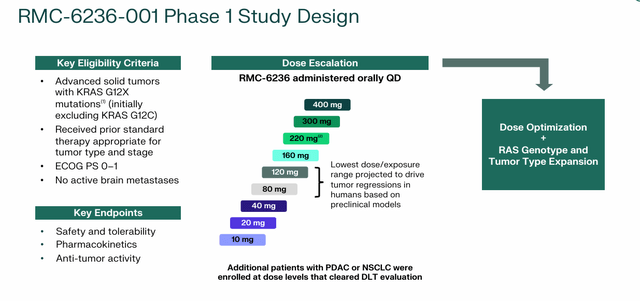
After showing promising results in human models in the clinic, the triple-compound RAS has been used orally once dailyMulti The inhibitor produced strong data across most KRAS G12 variants (referred to as “X,” meaning variants A, D, R, S, or V – not C) as monotherapy in a phase 1/1b dose-escalation study of 131 patients (RMC -6236-001). In 46 previously treated pancreatic ductal adenocarcinoma (PDAC) patients evaluated, RMC-6236 doses of 80 mg and higher resulted in a partial response (PR) or stable disease (SD) in 40 patients (87%). An objective response rate (ORR) of 20% and a disease control rate (DCR) of 67% compared favorably with the often used second-line (2L) combination chemotherapy of gemcitabine plus nab-paclitaxel (GnP; ORR 11%, DCR 56%). ). In 40 previously treated evaluable non-small cell lung cancer (NSCLC) patients, it produced 1 complete response (CR), 14 PRs, and 19 SDs for an ORR of 38% and a DCR of 85%, favorably compared with the standard of care for chemotherapy. Docetaxel (ORR 13%, DCR 60%). As for safety, there was 1 (of 131) grade 4 treatment-related adverse events (TRAE), with rash, nausea, diarrhea and vomiting the most common side effects. A further update to this ongoing study, which will include durability data, is expected in the second half of 2024.
However, the market, holding high expectations, initially responded as if it had a TRAE to the seemingly positive data, selling RVMD shares down 40% to $18.35 over two days in October 2023.
Based on these results, Revolution aims to initiate two registrational trials of RMC-6236 by YE24. One trial will evaluate treatment in more than 500 previously treated PDAC patients (about 85% of whom will harbor KRAS G12X variants) against GnP or Folfirinox chemotherapy agents with progression-free survival and overall survival on the proposed primary endpoints. The other is a similar study except the patient size (400+), indication (NSCLC), comparator (docetaxel), and KRAS G12X population will include a small but meaningful proportion of KRAS G12C patients.
RMC-6236 is also being evaluated in combination with Merck (MRK) The PD-1 inhibitor Keytruda (pembrolizumab) with or without chemotherapy in NSCLC patients as first-line treatment in a phase 1 trial (RMC-LUNG-101) with Revolution planning another early-stage chemotherapy combination study for 1L PDAC. Preliminary data from RMC-LUNG-101 is expected at 2H24.
RMC-6291. While RMC-6236 targets a broad range of KRAS G12 variants, Revolution’s RMC-6291 program focuses on selective KRAS G12C (ON) inhibition to avoid wild-type toxicity. Approximately 29,000 cases of KRAS G12C cancer—a hallmark of cigarette smoke—are diagnosed in the United States each year, with approximately 76% being lung cancer and 18% being colorectal cancer. First generation KRAS G12C inhibitors e.g Bristol Myers Squibb (BMY) Krazati (adagrasib) targets the OFF switch. After demonstrating superiority to Krazati in mouse models, RMC-6236 has entered the clinic in a Phase 1/1b dose-expansion study in previously treated solid KRAS G12C tumors (RMC-6291-001) with very encouraging initial yields.
May 2024 Company Presentation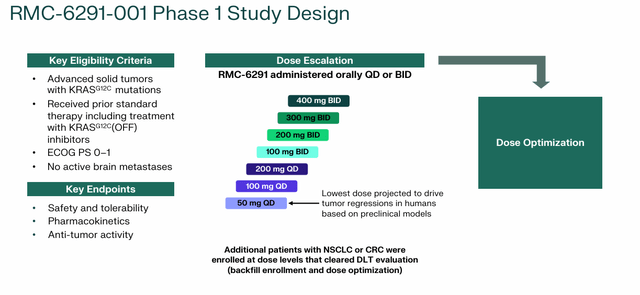
In 17 evaluable NSCLC patients, RMC-6291 achieved an ORR of 47% and a DCR of 100%. In 20 evaluable colorectal cancer (CRC) patients, the test produced an ORR of 40% and a DCR of 80%. The dosing trial is still ongoing. It is also being evaluated in the RMC-LUNG-101 combination study, and is scheduled for initial data acquisition at 1H25. RMC-6291 is also being evaluated in combination with RMC-6236 for the treatment of solid tumors.
RMC-9805. The company’s other selective RAS(ON) clinical program is RMC-9805, which targets KRAS G12D(ON) cancers, which number approximately 61,000 annually in the United States, of which PDAC, CRC, and NSCLC account for 89%. It is undergoing a phase 1 dose-finding monotherapy study and will be evaluated in combination with RMC-6236, both in solid tumors. Preliminary data is expected to be released from the first at 1H25 and the last at 2H24.
May 2024 Company Presentation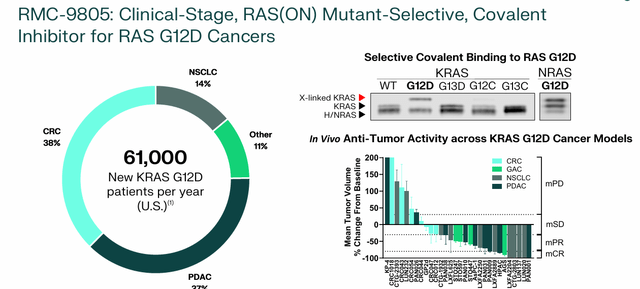
RMC-5552. Revolution has another clinical asset, the concomitant RAS inhibitor RMC-5552, which is designed to selectively inhibit mTORC1/4EBP1 while suppressing resistance to RAS inhibition by blocking cooperating mTORC1 pathway signaling. The intravenously administered drug candidate is being evaluated as monotherapy against multiple solid tumors in a phase 1/1b trial, with a DCR of 78% in 36 evaluable patients to date. However, this treatment does not currently appear to be a priority for management – likely due to bandwidth limitations. The same can be said for the companion inhibitor RMC-4630, which was returned by Sanofi (SNY) In June 2023; As well as the mutation-selective RAS (ON) inhibitors RMC-5127 (G12V), RMC-0708 (Q61H), and RMC-8839 (G13C).
With no assets currently leveraged, the company expects to market domestically while seeking business partnerships internationally, assuming approval(s). This wholly owned portfolio was likely a contributing factor to Wells Fargo’s decision to put Revolution on its list of potential acquisition targets the week of May 20.
Balance sheet and analyst comments:
In November 2023, the company essentially took approximately $1.1 billion of cash and securities off the balance sheet of EQRx’s failed “Global Drug Buyers Club” by issuing 54.8 million shares of RVMD. The purchase was agreed upon on July 31, 2023, and closed on November 9, 2023. Given the timing of the market’s reaction to the RMC-6236 data in October, Revolution ended up issuing approximately 18 million more shares than it had planned for a month. before. Either way, as of March 31, 2024, the company had $1.7 billion in cash and marketable securities, providing it with an operating opportunity. Cash runway In 2027.
Since Bank of America’s January 2024 upgrade, Street analysts have been unanimously positive on Revolution, boasting eight buy ratings, four outperform ratings, and an average price target of $45.
Verdict:
As for the competition, the company has listed at least 20 KRAS G12C (OFF) programs in the clinic and four more targeting KRAS G12D. Besides for Bridge Bio Pharma (BBIO), which has a selective KRAS G12C (ON) program, Revolution is truly the only biopharmaceutical concern targeting those proteins with ON therapeutics. The space is hot because the opportunity in solid tumors is great. I’m coming to update on BridgeBio Pharma earlier this week.
As such, RVMD shares have essentially doubled since the visceral reaction to the RMC-6236 clinical update seven months ago. This response was a result of the stock being perfectly priced. With a market capitalization of ~$4.5 billion net cash and the Street analysts’ quite positive average price target less than 20% away, Revolution stock is close to achieving price-for-perfection status once again, except one suitor thinks differently. With RMC-6236 durability data a potential fly in the ointment, now may be a good time to take at least some profits and wait for a better re-entry point if you currently own Revolution Medicines, Inc. shares.





